
Difference Between Oxidative and Nonoxidative Deamination Compare the Difference Between
It may be accomplished oxidatively or nonoxidatively. Oxidative deamination is stereospecific and is catalyzed by L- or D-amino acid oxidase. The initial step is removal of two hydrogen atoms by the flavin coenzyme, with formation of an unstable α-amino acid intermediate.
The Oxidative Deamination of Amino Acids docx D. Lamees Muhadharaty
May 8, 2023 by Vivek Kumar Oxidative and Non- Oxidative Deamination Contents: Introduction Oxidative deamination Non-oxidative deamination Difference between oxidative and non-oxidative deamination. Introduction The removal of amino group from the amino acid as ammonia (NH 3) is called deamination.

PPT Chapter 7 Catabolism of Proteins PowerPoint Presentation, free download ID599911
Non-oxidative Deamination In nonoxidative deamination, the amine group is removed without the oxidation process. A byproduct of non oxidative deamination is ammonia, producing consequent a-keto acids. Hydroxyl acids with one or more hydroxyl groups undergo non oxidative deamination.

🐈 Transamination and deamination. What is Deamination? (with pictures). 20190118
Oxidative deamination is a form of deamination that generates α-keto acids and other oxidized products from amine-containing compounds, and occurs primarily in the liver. [1]

PPT Amino acid metabolism PowerPoint Presentation, free download ID3816377
Figure 7.5.15 7.5. 15. Depurination of guanines (or adenines) is a common DNA lesion. Three of the four DNA bases, adenine, guanine, and cytosine, contain amine groups that can be lost in a variety of pH and temperature-dependent reactions that convert the bases to hypoxanthine, xanthine, and uracil, respectively.

PPT AMINO ACID METABOLISM PowerPoint Presentation, free download ID7122612
#deamination #oxidation #aminoacids #metabolismDeamination is the removal of an amino group from a molecule. Enzymes that catalyse this reaction are called d.

PPT Chapter 7 Catabolism of Proteins PowerPoint Presentation, free download ID599911
Amino acid metabolism lecture on Nonoxidative deamination.http://shomusbiology.com/Download the study materials here-http://shomusbiology.com/bio-materials.h.
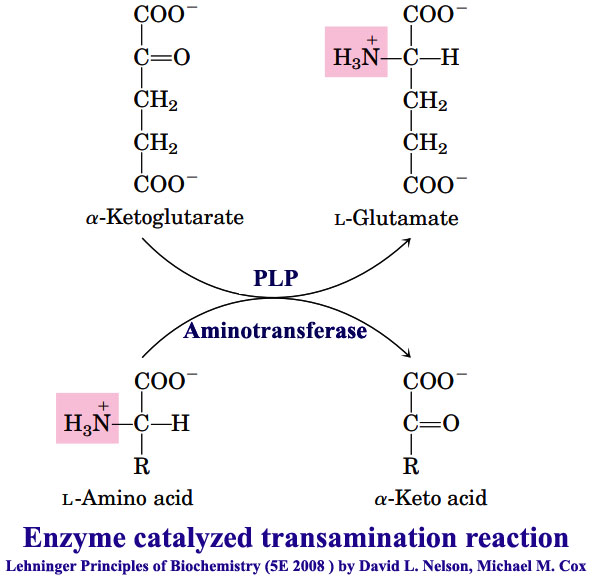
General reactions of amino acid metabolism Transamination, Oxidative and Nonoxidative
This video talks about Oxidative Deamination and amino acid catabolism.Also follow me on other social media -Facebook: https://www.facebook.com/arpan.parich..

PPT Protein metabolism PowerPoint Presentation, free download ID3540412
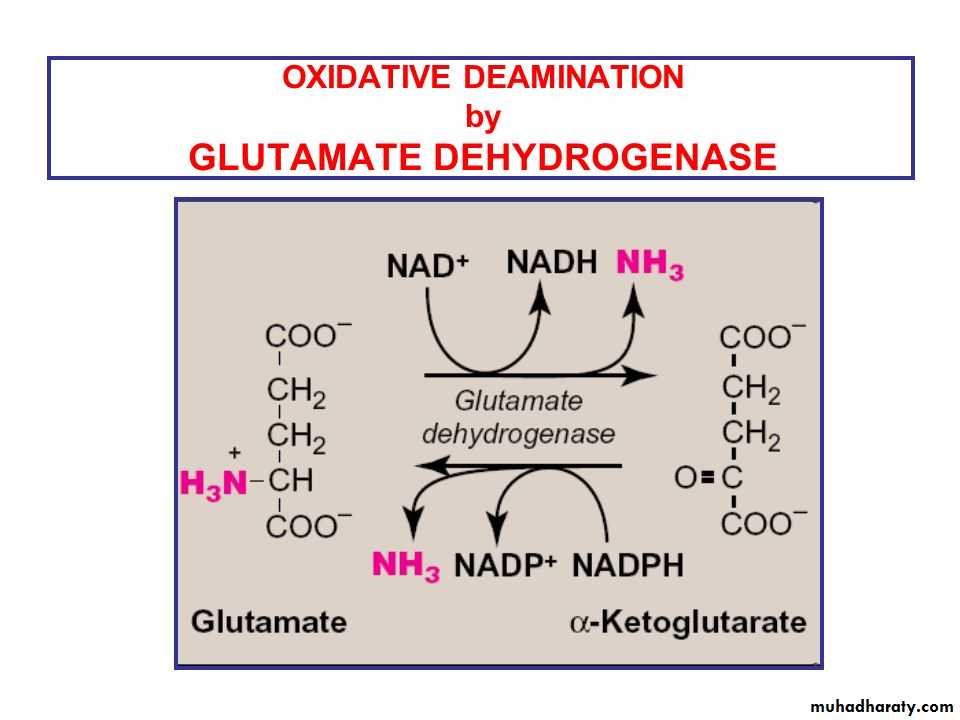
Glutamate is the only amino acid that undergoes oxidative deamination to a significant extent to liberate free NH3 for urea synthesis. All amino acids

Non Oxidative Deamination Reactions YouTube
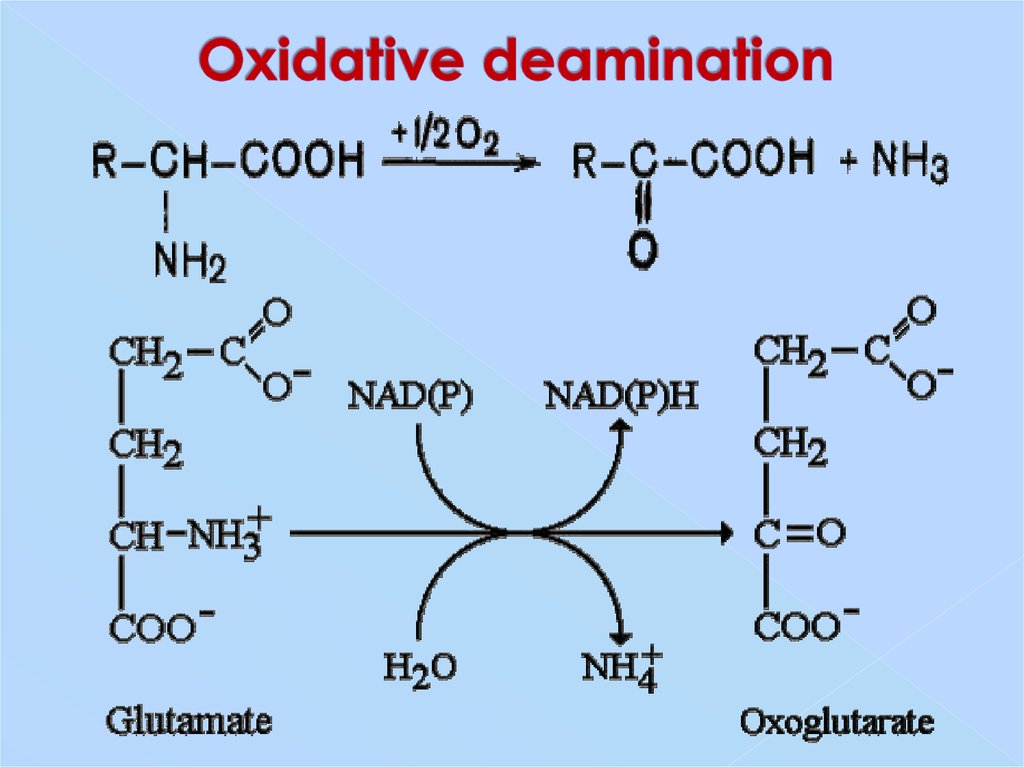
Oxidative Deamination. In the breakdown of amino acids for energy, the final acceptor of the α-amino group is α-ketoglutarate, forming glutamate. Glutamate can then undergooxidative deamination, in which it loses its amino group as an ammonium (NH 4 +) ion and is oxidized back to α-ketoglutarate (ready to accept another amino group):
Protein and amino acid metabolism online presentation
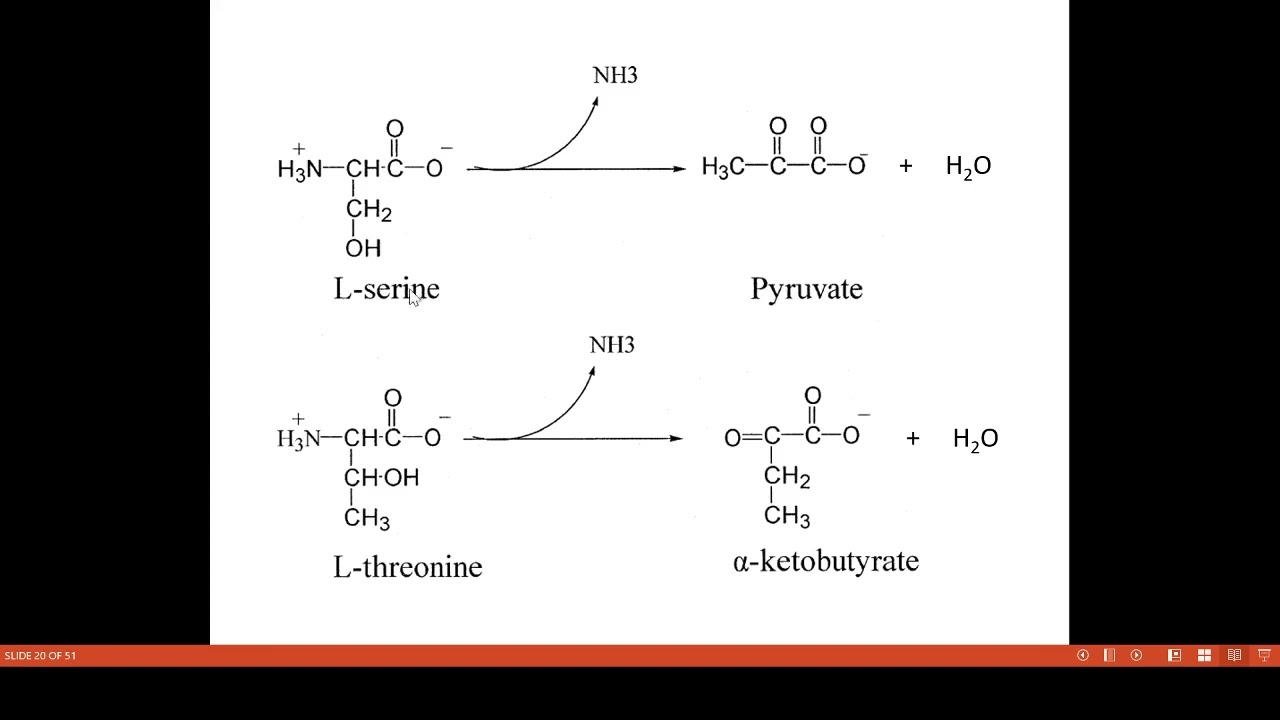
Nonoxidative deamination is a type of deamination reaction in which the removal of the amine group occurs without proceeding through an oxidation reaction. However, this type of deamination reactions liberates ammonia, producing the corresponding α-keto acids.
Video 7 non oxidative deamination YouTube
An overview of the oxidative deamination of N -acetylneuraminic acid derivatives (Neu5Ac) leading to the formation of ketodeoxynonulosonic acid (KDN), its stereoisomers and glycosides is presented.

PPT Protein metabolism PowerPoint Presentation, free download ID3540412
Oxidative Deamination. In the breakdown of amino acids for energy, the final acceptor of the α-amino group is α-ketoglutarate, forming glutamate. Glutamate can then undergooxidative deamination, in which it loses its amino group as an ammonium (NH 4 +) ion and is oxidized back to α-ketoglutarate (ready to accept another amino group):

What is the Difference Between Oxidative and Nonoxidative Deamination
3.1.2 Non-oxidative Deamination. It is the deamination of amino acids in the absence of molecular oxygen in cells. Non-oxidative deamination takes place by substrate-specific enzymes. Depending on types of amino acids, non-oxidative deamination has the following three types:

Deamination Oxidative and nonoxidative deamination YouTube
Nonoxidative deamination is a type of deamination reaction in which the removal of the amine group occurs without proceeding through an oxidation reaction. H.

General Reactions involved in amino acid metabolism
Oxidative deamination. Oxidative deamination reaction replaces −NH+3 − NH 3 + with a C=O C = O, producing an α α -keto acid and an ammonium ( NH+4 NH 4 +) ion, as shown in reaction 2 in Figure 9.7.1 9.7. 1. This is an oxidation reaction that is coupled with the reduction of NAD+ NAD + to NADH NADH.